About PD Partners
For more than 20 years, we’ve helped dozens of businesses in the highly-regulated GMP (good manufacturing practice) environment of drug development and testing. Our team of scientists is responsible for the development and refinement of new products. We provide ongoing testing for your drugs in ICH conditions and make sure that they meet all standards and parameters established by their stability date.
Integrity, communication, and sound science — these core values drive us in our research and ensure that we always do right by you (and ourselves). When you work with PD Partners, you can ensure you’ll get the best quality results. We focus on all of the details that often trip up businesses in the new product development process and provide guidance to help navigate them. From planning and formulation to testing and filing, we’ve got you covered.
Located in Tampa, Florida, our facility has 15,000 square feet of space with an adjacent unit of another 15,000 square feet. Our pristine production site has received all applicable environmental, health, and safety inspections as well as regulatory inspections to ensure it’s properly suited for your needs. We offer a full range of USP/BP/EP testing, ICH stability programs, product development, and regulatory consulting services to eliminate hiccups in the product development process.
Driven By Experience
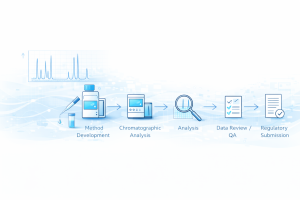
With more than 100 years of combined experience, our leadership team knows the intricacies that it takes to develop a new product, file it correctly, get approvals, and meet ever-changing government regulations. From pharmaceuticals to chemistry to microbiology, our team comes from all areas of scientific expertise. They oversee all operations — including managing formulation activities, document quality control, and cGMP oversight.
Providing Support for Your Industry
Developing a new product is no easy feat — not only do you need to ensure it’s made with the best quality ingredients, but it should also be safe for consumers, be approved by the FDA, and follow other strict government regulations. There are too many details to count. But, fortunately, you won’t have to. PD Partners takes care of the intricacies of new product development so that you can focus on making high-level decisions instead of getting caught in the weeds.
See What Industries We Support

Breaking New Ground
From the extensive development process to stability testing, validation services, and FDA submissions, our services help you navigate the intricate and often complex process of new drug development. We’ll help you avoid mistakes that often derail projects and streamline the process to eliminate financial pitfalls — helping you meet strict deadlines. No matter your needs during the development process, PD Partners is here to help you.

